Diversity of Secondary Structure in Catalytic Peptides with β-Turn-Biased Sequences | Journal of the American Chemical Society

NiH-catalyzed anti-Markovnikov hydroamidation of unactivated alkenes with 1,4,2-dioxazol-5-ones for the direct synthesis of N-alkyl amides | Communications Chemistry

Frank MEYSKENS | Professor of Medicine, Biological Chemistry, Epidemiology and Public Health | University of California, Irvine, CA | UCI | Department of Medicine | Research profile

Cysteine‐Selective Modification of Peptides and Proteins via Desulfurative C−C Bond Formation - Griffiths - Chemistry – A European Journal - Wiley Online Library

Enantioselective Bromolactonization of Conjugated (Z)-Enynes | Journal of the American Chemical Society
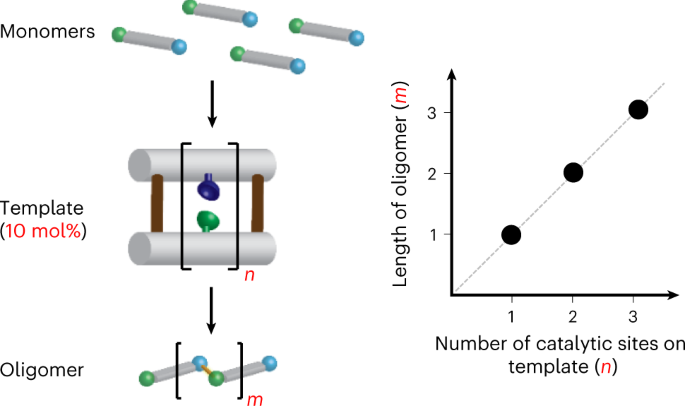
Catalytic length-controlled oligomerization with synthetic programmable templates | Nature Synthesis

41BHQ2vi-JL._CLa|603,500|5151D4ePjDL.jpg,51PQQUIZhjL.jpg|0,0,274,500+329,0,274,500+137,0,329,500__._SX400_SY326_.jpg